batteryP2DResults
Description
A batteryP2DResults object contains the
concentration of Li-ion and electric potential in solid active material particles in both the
anode and cathode, as well as in the liquid electrolyte. The object also contains the voltage
at the battery terminals, ionic flux, solution times, and mesh.
Creation
Solve a battery pseudo-2D (P2D) problem using the solve function. The solution returned by this function is a
batteryP2DResults object.
Properties
This property is read-only.
Solution times, represented as a numeric vector. The time is specified in seconds (s).
Data Types: double
This property is read-only.
Voltage at the battery terminals, represented as a numeric vector. The voltage is specified in Volts (V).
Data Types: double
Rate at which the battery is charged or discharged relative to its ideal capacity, represented as a numeric vector.
Data Types: double
This property is read-only.
Concentration of Li-ion in the liquid electrolyte, represented as a numeric matrix whose rows correspond to the solution times, and columns correspond to the battery mesh nodes. The concentration is specified in moles per cubic meter (mol/m3).
Data Types: double
This property is read-only.
Electric potential in the solid phase in both the anode and cathode, represented as
a numeric matrix whose rows correspond to the solution times, and columns correspond to
the battery mesh nodes. Matrix elements corresponding to the nodes in the separator
region are NaN. The potential is specified in Volts (V).
Data Types: double
This property is read-only.
Electric potential in the liquid electrolyte, represented as a numeric matrix whose rows correspond to the solution times, and columns correspond to the battery mesh nodes. The potential is specified in Volts (V).
Data Types: double
This property is read-only.
Li-ion flux at the interface of solid and liquid phase, represented as a numeric
matrix whose rows correspond to the solution times, and columns correspond to the
battery mesh nodes. Matrix elements corresponding to the nodes in the separator region
are NaN. The Li-ion flux is specified in moles per square meter per
second (mol/(m2s)).
Data Types: double
This property is read-only.
Average concentration of Li-ion in the solid active material particles in both the
anode and cathode, represented as a numeric matrix whose rows correspond to the solution
times, and columns correspond to the battery mesh nodes. Averaging is performed along
the radial direction of a particle at each mesh node. Matrix elements corresponding to
the nodes in the separator region are NaN. Average concentration is a
normalized value, and therefore, it is unitless.
Data Types: double
This property is read-only.
Concentration of lithium ions at the particle radius surface, represented as a
numeric matrix whose rows correspond to the solution times, and columns correspond to
the battery mesh nodes. This value is the SolidConcentration value
at r =
RP, where RP is the particle
radius. Matrix elements corresponding to the nodes in the separator region are
NaN. Surface solid concentration is a normalized value, and
therefore, it is unitless.
Data Types: double
This property is read-only.
Concentration of Li-ion in the solid active material particles in both the anode
and cathode, represented as a 3-D numeric array. The first dimension corresponds to the
solution times, the second dimension corresponds to the distances along the particle
radius, and the third dimension correspond to the battery mesh nodes. Array elements
corresponding to the nodes in the separator region are NaN. Solid
concentration is a normalized value, and therefore, it is unitless.
Data Types: double
This property is read-only.
Battery mesh, returned as a batteryMesh
object.
Object Functions
plotSummary | Plot battery P2D modeling solution |
Examples
Solve a Li-Ion battery electrochemistry problem by using a pseudo-2D battery model.
Both the anode and cathode materials require the open circuit potential specification, which determines the voltage profile of the battery during charging and discharging. The open circuit potential is a voltage of electrode material as a function of the stoichiometric ratio, which is the ratio of intercalated lithium in the solid to maximum lithium capacity. You can specify this ratio by interpolating the gridded data set.
sNorm = linspace(0.025, 0.975, 39); ocp_n_vec = [.435;.325;.259;.221;.204; ... .194;.179;.166;.155;.145; ... .137;.131;.128;.127;.126; ... .125;.124;.123;.122;.121; ... .118;.117;.112;.109;.105; ... .1;.098;.095;.094;.093; ... .091;.09;.089;.088;.087; ... .086;.085;.084;.083]; ocp_p_vec = [3.598;3.53;3.494;3.474; ... 3.46;3.455;3.454;3.453; ... 3.4528;3.4526;3.4524;3.452; ... 3.4518;3.4516;3.4514;3.4512; ... 3.451;3.4508;3.4506;3.4503; ... 3.45;3.4498;3.4495;3.4493; ... 3.449;3.4488;3.4486;3.4484; ... 3.4482;3.4479;3.4477;3.4475; ... 3.4473;3.447;3.4468;3.4466; ... 3.4464;3.4462;3.4458]; anodeOCP = griddedInterpolant(sNorm,ocp_n_vec,"linear","nearest"); cathodeOCP = griddedInterpolant(sNorm,ocp_p_vec,"linear","nearest");
Create objects that specify the active materials for the anode and cathode.
anodeMaterial = batteryActiveMaterial( ... ParticleRadius=5E-6, ... MaximumSolidConcentration=30555, ... VolumeFraction=0.58, ... DiffusionCoefficient=3.0E-15, ... ReactionRate=8.8E-11, ... OpenCircuitPotential=@(st_ratio) anodeOCP(st_ratio), ... StoichiometricLimits=[0.0132 0.811]); cathodeMaterial = batteryActiveMaterial(... ParticleRadius=5E-8, ... MaximumSolidConcentration=22806, ... VolumeFraction=0.374, ... DiffusionCoefficient=5.9E-19, ... ReactionRate=2.2E-13, ... OpenCircuitPotential=@(st_ratio) cathodeOCP(st_ratio), ... StoichiometricLimits=[0.035 0.74]);
Next, create objects that specify both electrodes.
anode = batteryElectrode(... Thickness=34E-6, ... Porosity=0.3874, ... BruggemanCoefficient=1.5, ... ElectricalConductivity=100, ... ActiveMaterial=anodeMaterial); cathode = batteryElectrode(... Thickness=80E-6, ... Porosity=0.5725, ... BruggemanCoefficient=1.5, ... ElectricalConductivity=0.5, ... ActiveMaterial=cathodeMaterial);
Create an object that specifies the properties of the separator.
separator = batterySeparator(... Thickness=25E-6, ... Porosity=0.45, ... BruggemanCoefficient=1.5);
Create an object that specifies the properties of the electrolyte.
electrolyte = batteryElectrolyte(... DiffusionCoefficient=2E-10, ... TransferenceNumber=0.363, ... IonicConductivity=0.29);
Create an object that specifies the initial conditions of the battery.
ic = batteryInitialConditions(... ElectrolyteConcentration=1000, ... StateOfCharge=0.05, ... Temperature=298.15);
Create an object that specifies the properties of the battery cycling step.
cycling = batteryCyclingStep(... NormalizedCurrent=0.5, ... CutoffTime=100, ... CutoffVoltageUpper=4.2, ... OutputTimeStep=10);
Create a model for the battery P2D analysis.
model = batteryP2DModel(... Anode=anode, ... Separator=separator, ... Cathode=cathode, ... Electrolyte=electrolyte, ... InitialConditions=ic, ... CyclingStep=cycling);
Set the maximum step size for the internal solver to 2.
model.SolverOptions.MaxStep = 2;
Solve the model using the solve function. The resulting object contains the concentration of Li-ion and electric potential in solid active material particles in both the anode and cathode, as well as in the liquid electrolyte. The object also contains the voltage at the battery terminals, ionic flux, solution times, and mesh.
results = solve(model)
results =
batteryP2DResults with properties:
SolutionTimes: [11×1 double]
TerminalVoltage: [11×1 double]
NormalizedCurrent: [11×1 double]
LiquidConcentration: [11×49 double]
SolidPotential: [11×49 double]
LiquidPotential: [11×49 double]
IonicFlux: [11×49 double]
AverageSolidConcentration: [11×49 double]
SurfaceSolidConcentration: [11×49 double]
SolidConcentration: [11×7×49 double]
Mesh: [1×1 batteryMesh]
Visualize the results.
plotSummary(results)
![Figure contains 6 axes objects. Axes object 1 with title Terminal Voltage [V], xlabel Time [s] contains an object of type line. Axes object 2 with title Normalized Current, xlabel Time [s] contains an object of type line. Axes object 3 with title Liquid Concentration [mol/m Cubed baseline ], xlabel Thickness [m] contains 7 objects of type line, rectangle. Axes object 4 with title Normalized Average Solid Concentration, xlabel Thickness [m] contains 7 objects of type line, rectangle. Axes object 5 with title Liquid Potential [V], xlabel Thickness [m] contains 7 objects of type line, rectangle. Axes object 6 with title Solid Potential [V], xlabel Thickness [m] contains 7 objects of type line, rectangle. These objects represent 0, 30, 60, 100.](../../examples/pde/win64/SolveModelForBatteryP2DAnalysisExample_01.png)
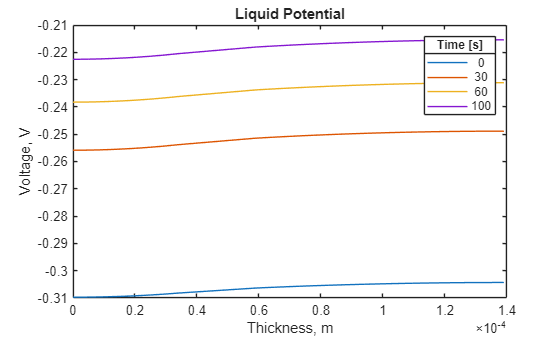
To see more details of the liquid potential distribution, plot it separately. Use the same four solution times.
figure for i = [1 4 7 11] plot(results.Mesh.Nodes, ... results.LiquidPotential(i,:)) hold on end title("Liquid Potential") xlabel("Thickness, m") ylabel("Voltage, V") lgd = legend(num2str(results.SolutionTimes([1;4;7;11]))); lgd.Title.String = "Time [s]";
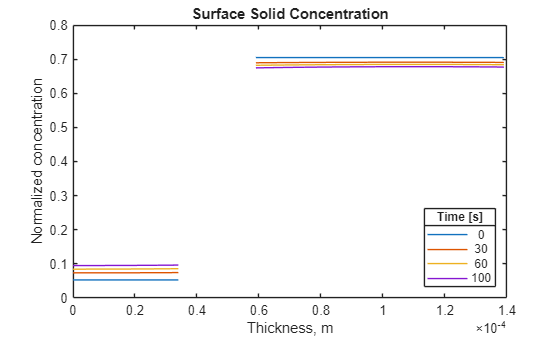
Plot the surface solid concentration distribution for the same four solution times.
figure for i = [1 4 7 11] plot(results.Mesh.Nodes, ... results.SurfaceSolidConcentration(i,:)) hold on end title("Surface Solid Concentration") xlabel("Thickness, m") ylabel("Normalized concentration") lgd = legend(num2str(results.SolutionTimes([1;4;7;11]))); lgd.Location = "southeast"; lgd.Title.String = "Time [s]";
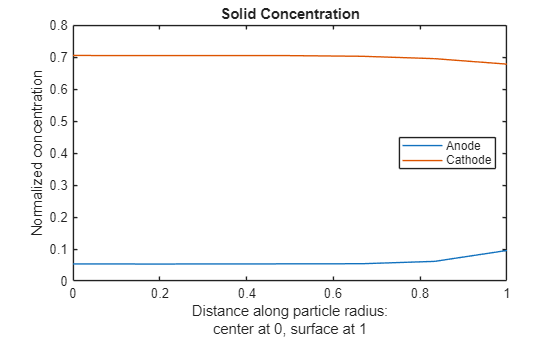
For the final solution time, 100s, plot the solid concentration along the particle radius at two locations corresponding approximately to the middle of the anode and the middle of the cathode.
figure Rfinal = results.SolidConcentration(end,:,:); Nr = size(results.SolidConcentration,2); for x = [results.Mesh.AnodeNodes(ceil(end/2)) ... results.Mesh.CathodeNodes(ceil(end/2))] Rr = Rfinal(:,:,x); plot((0:Nr-1).'/(Nr-1),Rr(:)) hold on end title("Solid Concentration") xlabel({"Distance along particle radius:";"center at 0, surface at 1"}) ylabel("Normalized concentration") legend("Anode","Cathode",Location="east");
Model the battery discharging for four different constant values of the normalized current.
Specify the universal gas constant, the Faraday constant, the Boltzmann constant, the elementary charge, and the simulation temperature.
R = 8.31446; % J/(mol·K)) FaradayConstant = 96487; % C/mol BoltzmannConstant = 1.380649E-23; % J/K ElementaryCharge = 1.6021766E-19; % C SimulationTemperature = 298.15; % K
The open circuit potential is a voltage of electrode material as a function of the stoichiometric ratio, which is the ratio of intercalated lithium in the solid to maximum lithium capacity. Create this function to specify the open circuit potential of the anode.
function anodeOCP = anodefuncOCP(st_ratio) anodeOCP = -591.7*st_ratio.^9+2984*st_ratio.^8 ... -6401*st_ratio.^7+7605*st_ratio.^6- ... 5465*st_ratio.^5+2438*st_ratio.^4 ... -670.5*st_ratio.^3+110.2*st_ratio.^2- ... 10.39*st_ratio+0.6363; end
Specify the properties of the battery anode by creating a batteryActiveMaterial object. The properties of this object specify the active material of the anode, including the particle radius, maximum solid-phase concentration of lithium ions, fraction of the anode volume occupied by the active material, diffusion coefficient, reaction rate, open circuit potential of the anode material as a function of the stoichiometric ratio, and the range of stoichiometric values.
anodeMaterial = batteryActiveMaterial( ... ParticleRadius=5E-6, ... VolumeFraction=0.58, ... MaximumSolidConcentration=30555, ... DiffusionCoefficient=3.0E-15, ... ReactionRate=8.8E-11, ... OpenCircuitPotential=@anodefuncOCP, ... StoichiometricLimits=[0.0132 0.811]);
Specify the open circuit potential of the cathode by interpolating the gridded data set.
sNorm = linspace(0.025, 0.975, 39); ocp_p_vec = [3.598;3.53;3.494;3.474; ... 3.46;3.455;3.454;3.453; ... 3.4528;3.4526;3.4524;3.452; ... 3.4518;3.4516;3.4514;3.4512; ... 3.451;3.4508;3.4506;3.4503; ... 3.45;3.4498;3.4495;3.4493; ... 3.449;3.4488;3.4486;3.4484; ... 3.4482;3.4479;3.4477;3.4475; ... 3.4473;3.447;3.4468;3.4466; ... 3.4464;3.4462;3.4458]; cathodeOCP = griddedInterpolant(sNorm,ocp_p_vec,"linear","nearest");
Specify the diffusion coefficient as a function of the solid concentration.
DiffusivityReference = @(s)5.9E-19*((s-0.5).^2+0.75);
Create a batteryActiveMaterial object that specifies the active material of the cathode, including the particle radius, maximum solid-phase concentration of lithium ions, fraction of the anode volume occupied by the active material, diffusion coefficient, reaction rate, voltage of the anode material as a function of the stoichiometric ratio, and the range of stoichiometric values.
cathodeMaterial = batteryActiveMaterial( ... ParticleRadius=5E-8, ... VolumeFraction=0.374, ... MaximumSolidConcentration=22806, ... DiffusionCoefficient= @(r,s) DiffusivityReference(s.SolidConcentration/22806), ... ReactionRate=2.2E-13, ... OpenCircuitPotential=@(st_ratio) cathodeOCP(st_ratio), ... StoichiometricLimits=[0.035 0.74]);
Create the objects that specify the anode and cathode properties, such as the thickness, porosity, Bruggeman's coefficient, electrical conductivity, and active material.
anode = batteryElectrode( ... Thickness=34E-6, ... Porosity=0.3874, ... BruggemanCoefficient=1.5, ... ElectricalConductivity=100, ... ActiveMaterial=anodeMaterial); cathode=batteryElectrode( ... Thickness=80E-6, ... Porosity=0.5725, ... BruggemanCoefficient=1.5, ... ElectricalConductivity=0.5, ... ActiveMaterial=cathodeMaterial);
Specify the thickness, porosity, and Bruggeman's coefficient of the battery separator.
separator=batterySeparator( ... Thickness=25E-6, ... Porosity=0.45, ... BruggemanCoefficient=1.5);
Specify the diffusion coefficient, transference number, and ionic conductivity of the battery electrolyte.
ConductivityElectrolyte = ... @(ce) -4.582e-01*(ce/1000).^2 + 1.056*(ce/ 1000) + 0.3281; electrolyte = batteryElectrolyte( ... DiffusionCoefficient= ... @(r,s) BoltzmannConstant/(FaradayConstant*ElementaryCharge)* ... SimulationTemperature* ... ConductivityElectrolyte(s.LiquidConcentration)./s.LiquidConcentration, ... TransferenceNumber=0.363, ... IonicConductivity=@(x,s) ConductivityElectrolyte(s.LiquidConcentration));
Specify the minimum voltage limit for the battery during discharging. Also specify the output time step for the solver.
cycling = batteryCyclingStep( ... CutoffVoltageLower=2.8, ... OutputTimeStep=1);
Specify the initial temperature for the battery.
ic = batteryInitialConditions( ...
Temperature=SimulationTemperature);Create a model for the battery P2D analysis and set its properties using the objects created.
model = batteryP2DModel( ... Anode=anode, ... Cathode=cathode, ... Separator=separator, ... Electrolyte=electrolyte, ... CyclingStep=cycling, ... InitialConditions=ic);
Specify the state of charge (SoC) as 1 to indicate that the battery is fully charged.
model.InitialConditions.StateOfCharge = 1;
Specify the electrolyte concentration for the fully charged battery.
model.InitialConditions.ElectrolyteConcentration = 1200;
Set the maximum step size for the internal solver to 2.
model.SolverOptions.MaxStep = 2;
Solve the model for four values of the normalized current: –0.2, –0.5, –1, and –2. Negative values indicate that the battery is discharging.
model.CyclingStep.NormalizedCurrent = -0.2; R02 = solve(model); model.CyclingStep.NormalizedCurrent = -0.5; R05 = solve(model); model.CyclingStep.NormalizedCurrent = -1; R1 = solve(model); model.CyclingStep.NormalizedCurrent = -2; R2 = solve(model);
Plot the resulting terminal voltage at each solution time.
plot(R02.SolutionTimes./3600,R02.TerminalVoltage) hold on plot(R05.SolutionTimes./3600,R05.TerminalVoltage) plot(R1.SolutionTimes./3600,R1.TerminalVoltage) plot(R2.SolutionTimes./3600,R2.TerminalVoltage) xlabel("Time,hours") ylabel("Voltage,V") legend("0.2C","0.5C","1C","2C"); hold off

Version History
Introduced in R2026a
See Also
Objects
batteryP2DModel|batteryElectrode|batteryElectrolyte|batterySeparator|batteryActiveMaterial|batteryCyclingStep|batteryInitialConditions|batteryMesh|batterySolverOptions
Functions
MATLAB Command
You clicked a link that corresponds to this MATLAB command:
Run the command by entering it in the MATLAB Command Window. Web browsers do not support MATLAB commands.
Seleziona un sito web
Seleziona un sito web per visualizzare contenuto tradotto dove disponibile e vedere eventi e offerte locali. In base alla tua area geografica, ti consigliamo di selezionare: .
Puoi anche selezionare un sito web dal seguente elenco:
Come ottenere le migliori prestazioni del sito
Per ottenere le migliori prestazioni del sito, seleziona il sito cinese (in cinese o in inglese). I siti MathWorks per gli altri paesi non sono ottimizzati per essere visitati dalla tua area geografica.
Americhe
- América Latina (Español)
- Canada (English)
- United States (English)
Europa
- Belgium (English)
- Denmark (English)
- Deutschland (Deutsch)
- España (Español)
- Finland (English)
- France (Français)
- Ireland (English)
- Italia (Italiano)
- Luxembourg (English)
- Netherlands (English)
- Norway (English)
- Österreich (Deutsch)
- Portugal (English)
- Sweden (English)
- Switzerland
- United Kingdom (English)